|
3/20/2024 0 Comments Periodic table reactivity trend 
The repeating properties in each row of the periodic table, as observed by Mendeleev and others, reflect the repeating electron configurations in subsequent rows. The valence are also the highest-energy electrons in an atom, and most likely to participate in a reaction.įor these reasons, atoms with similar electron configurations generally behave in similar ways. That fact makes the valence electrons more likely to interact with other atoms. The valence electrons are the outermost electrons in an atom they are closest to the surface of an atom. One atom may donate electrons to another atoms. In a reaction, one atom may accept electrons from another atom. Atoms with similar electron configurations have similar properties.Ĭhemical reactions depend on the movement of electrons.The periodic table is divided into columns of atoms with similar electron configurations.The periodic table shown with the lanthanides in their proper places. 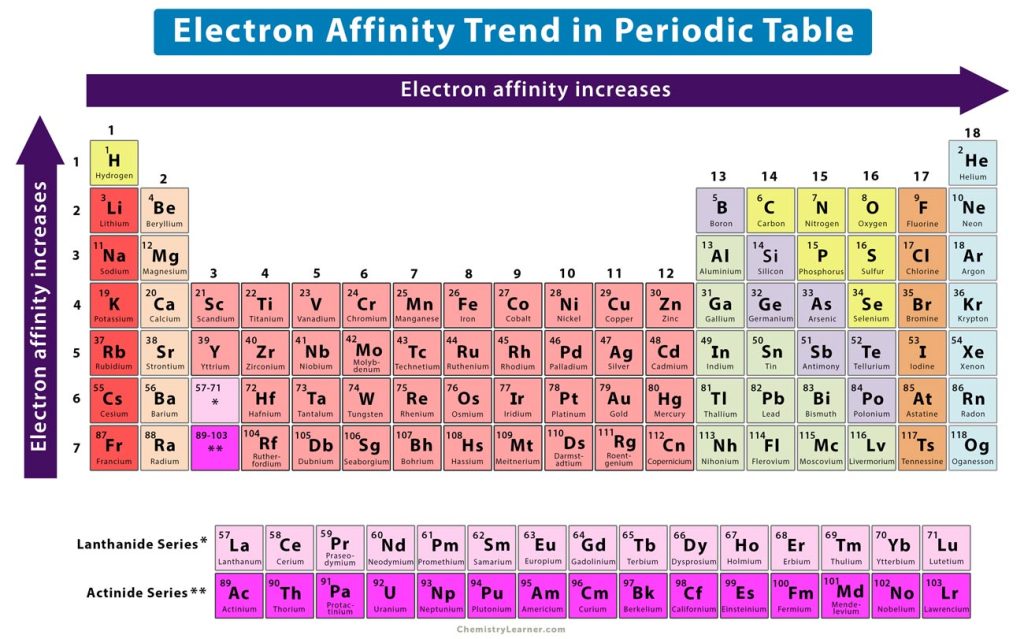
Really, the periodic table should look like this:įigure AT6.2. The f-block elements are usually shown below in order to save space. The element that really occurs next is element 58, cerium, and it is shown in the lanthanide row down below. Notice that lanthanum, element 57, is followed by hafnium, element 72, in the table. These elements could really be inserted at the left-hand side of the d-block in the appropriate rows. Collectively, they are called the f-block elements. The final two rows of the periodic table are the lanthanides and actinides. For example, scandium has configuration 4s 23d 1. The middle block of the periodic table consists of the transition metals or the d-block elements. For example, phosphorus has a configuration, 4s 24p x 1p y 1p z 1, or simply 4s 24p 3. Alternatively, they are sometimes called the s-block and p-block elements, respectively. The first two and the last six columns of the periodic table are called the main group elements. The main group is divided into metals (green), metalloids (teal), non-metals (blue) and noble or inert gases (purple). 
Green, blue and purple elements are the p-block together with the s-block, they are called the main group elements. Orange elements are the lanthanides and actinides (f-block). Yellow elements are the transition metals (d-block). Red elements are the alkali and alkaline earth metals (s-block). In the case of potassium, they are the ones beyond. Remember, the valence electrons are the ones beyond the noble gas core. 
Together, these elements are often called the s-block elements, because their valence electrons are s electrons. Alkali elements, from the first column, have a configuration ending in s 1 alkaline earth elements, from the second column, have configurations ending in s 2. These atoms are often called the alkali and alkaline earth elements. For example, potassium has a configuration 4s 1. If the periodic table is used as a tool, this process is pretty easy.įor atoms found in the first two columns of the periodic table (figure AT5.1), the configuration is a closed shell of core electrons, plus s electrons in a new shell. The aufbau process is a set of rules that allows us to predict the electronic configuration of an atom if we know how many electrons there are in the atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
